At a Glance
An at-home sleep study is a test that measures breathing patterns, oxygen levels, and other key metrics while you sleep in your own bed to help diagnose obstructive sleep apnea. These tests are more convenient and affordable than in-lab studies but collect fewer measurements and are typically recommended for people with a strong likelihood of moderate to severe sleep apnea.
Traditionally, to be diagnosed with sleep apnea, you had to complete a sleep study called a polysomnography (PSG) in a lab, hospital, or clinic. While in-lab testing is still the gold standard, an a-home sleep apnea test is another option that you and your doctor may decide is a good fit for you.
At-home sleep apnea tests, like the WatchPAT ONE At-Home Sleep Study, are best suited for people who are suspected of having moderate to severe obstructive sleep apnea (OSA) that isn’t complicated by other disorders. Taking a sleep apnea test at-home can be more convenient and comfortable than in a lab, but it may have limitations as well.




Sleep Doctor
An FDA-cleared, 98% accurate, disposable sleep apnea test that accurately measures key sleep metrics to diagnose you in the comfort of your own home.
Order, test, and get results, all from home
Take a diagnostic test that reveals your specific sleep issues
Get a personalized sleep improvement plan
Personalized
customer care
HSA/FSA
Eligible
Free & fast
shipping
Sleep Test Product Details
- Chest, wrist, and finger sensors measure peripheral arterial tone, heart rate, blood oxygen, actigraphy, body position, snoring, and chest motion.
- Testers receive an AHI (apnea hypoxia index) score in their follow-up email.
- Those who meet diagnostic criteria for OSA after testing may be prescribed PAP therapy.
- Unopened tests may be returned within 60 days.
How to Take the Test
- Fill out the assessment forms
- Book a quick online chat with a doctor
- The doctor will check if you might have OSA
- You’ll talk with a sleep coach about your options
- They’ll help you plan your next steps
- The test is single-use
- You must be 18 or older to take it
Shipping & Returns
Shipping is free within the continental United States (except for Alaska, Hawaii, Puerto Rico, Guam, Virgin Islands). The Sleep Doctor will not ship products internationally. All applicable shipping options will be displayed at time of checkout.
If you have ordered a Home Sleep Test device, the order will be sent through USPS Priority Mail and should reach you within 3 business days.
Return Policy:
We want you to be completely satisfied with your purchase, so we offer a 60-night return option from the date of delivery and a partial refund. To be eligible for a return, the product must be in new and unopened condition.
Please note that you are responsible for return shipping, and expedited shipping costs are non-refundable. Refunds may take up to 10 business days to process.
What Is an At-Home Sleep Study?
Also called an unattended sleep study or out-of-center sleep testing, an at-home sleep study typically involves wearing a small device that monitors your breathing, heart rate, oxygen levels, and other key metrics while you sleep. Specifically, they focus on detecting signs of obstructive sleep apnea (OSA), such as pauses in breathing or drops in oxygen.
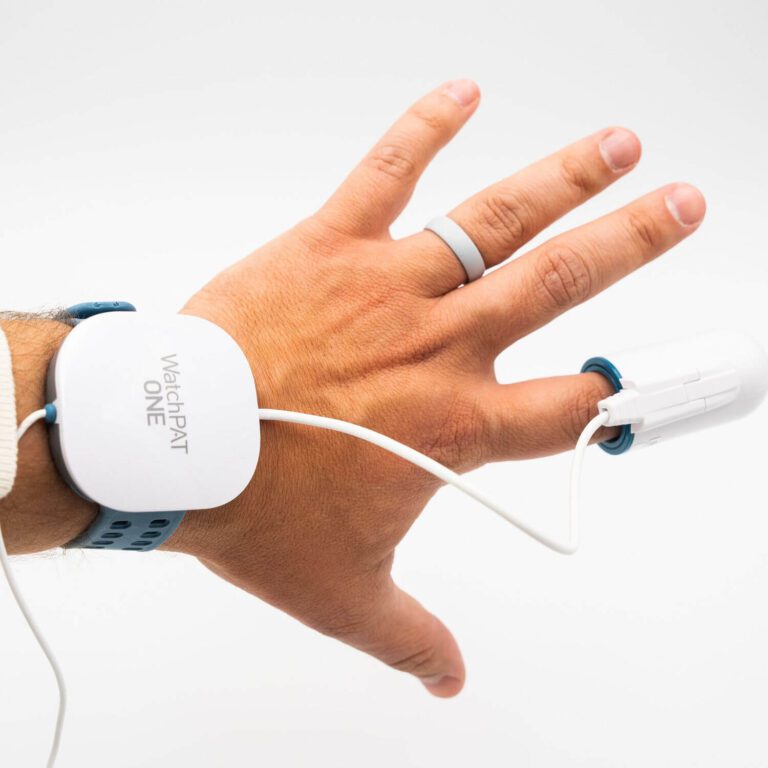
The WatchPAT ONE, for instance, is a wearable device that's worn on the fingertip and wrist, similar to a watch. The sensor on the fingertip measures blood oxygen levels and heart rate while the other sensor, which adheres to the chest, monitors breathing patterns and sleep positions throughout the night.
That being said, there are three different types of home sleep apnea devices, though types 3 and 4 are the most common.
- Type 2: These tests can measure the same data as an in-lab polysomnography, which may be called a type 1 test. A technician usually helps you set up the sensors or leads at a sleep center, or they may teach you how to do it at-home. Type 2 tests are not often used, because they can be too complex for a home setting.
- Type 3: These portable monitoring devices can measure up to seven different variables, including respiratory, cardiac, and blood oxygen measures. Unlike in-lab tests, type 3 tests can’t distinguish between wakefulness and sleep because they don’t measure brain activity. But some type 3 devices can estimate sleep measures by detecting your movement.
- Type 4: Type 4 devices are also portable but only measure one to three variables, like airflow and blood oxygen level.
What Does an At-Home Sleep Study Measure?
At-home sleep apnea testing measures certain body metrics like airflow, heart rate, blood oxygen, and snoring. These measures can demonstrate that your breathing is interrupted during sleep, which can help sleep specialists make a sleep apnea diagnosis. While at-home devices differ, these are the variables commonly measured:
- Respiratory effort: This is a measure of how well you breathe in and out. Some at-home sleep apnea tests use a belt placed around the chest to measure how your chest rises and falls as you breathe.
- Airflow: With some tests, you place a device called a nasal cannula inside each nostril to measure the flow of air into and out of your nose.
- Snoring: Some tests can detect snoring by using a chest sensor or nasal pressure sensor.
- Oxygen saturation: A device known as a pulse oximeter measures how much oxygen is in your blood. The device is usually placed on the tip of your finger.
- Heart rate: The pulse oximeter you wear on your finger typically also measures your heart rate.
- Body position and movement: Certain test devices include sensors that can detect your body position and movement.
- Peripheral arterial tone (PAT): PAT measures the change in volume of your finger artery, which corresponds to abnormal breathing events. Some at-home tests measure PAT as a way to track breathing, rather than measuring airflow or respiratory effort. These devices don’t require a chest band or nasal cannula.
Unlike PSG, type 3 and 4 at-home tests can’t directly measure sleep activity like sleep stages because they don’t use electrodes that can detect brain activity. These at-home tests focus on breathing and oxygen levels, though some rely on secondary measures, like body movement, to indicate sleep quality.
Your sleep specialist will interpret your results after your test. They will likely focus on one of two sleep apnea indices, depending on your specific device.
- Apnea-hypopnea index (AHI): The AHI is the number of breathing disruptions per hour of sleep.
- Respiratory event index (REI): The REI represents the number of breathing disruptions over the total testing time.
How Does an At-Home Sleep Study Work?
If you're experiencing sleep apnea symptoms, such as loud snoring or daytime sleepiness, there are several steps that can help you obtain an at-home test and take it successfully.
- Talk to your doctor: The first step is to talk to your doctor about your symptoms. Your doctor will likely ask questions about your personal health history and family history. Your doctor may also conduct a physical exam and assess factors such as your age, sex, and weight in order to determine your sleep apnea risk.
- Get a prescription: If your doctor finds you’re at risk for moderate to severe obstructive sleep apnea, they can submit an order for an at-home sleep apnea test. There are also options to order an at-home sleep apnea test online.
- Get your test: The test may be delivered to you, or you may go to a sleep study center to get your device and instructions on how to use it.
- Apply the sensors: When you’re ready to use the test, follow the instructions for your specific device to place the sensors on your body correctly.
- Turn on the device: Follow the instructions and turn on the device when you’re ready to sleep. Some devices also pair to a phone app.
- Sleep normally: Maintain your usual bedtime routine and try to go to sleep as you normally do each night, with the appropriate leads and sensors attached to your body. Most studies require one full night of sleep, including at least four hours of uninterrupted monitoring data.
- Remove the sensors: When you wake up, turn off the device according to the instructions and remove the sensors. Return the device if required.
- Discuss results with your doctor: Your doctor might make treatment recommendations based on your sleep study results, or they may determine that you need additional tests.
What Are the Pros of an At-Home Sleep Study?
- Easy access: It may be difficult for you to complete an in-lab sleep apnea test depending on where you live, scheduling constraints, and other responsibilities. Having the equipment brought to your home makes testing more accessible.
- Comfort: You may find the thought of sleeping in a foreign environment unappealing and prefer familiar surroundings instead.
- Flexibility: Having the flexibility to complete the test at-home when it works best for you may make you more likely to do it.
- Lower cost: At-home tests are usually more affordable than tests in labs because they don’t require technicians to be on hand. Some at-home tests may also be covered by insurance.
- Shorter wait: You can take the test at-home when you’re ready instead of when there’s an appointment available at a sleep center. You might be able to get treatment more quickly if you’re diagnosed sooner.
Here's how the WatchPAT One measures up:
- Affordable: It typically costs $189, but you can often catch sales and deals on its retail price.
- Offers telehealth care: If you're worried about reading and understanding your results, you'll be able to talk to a licensed sleep doctor both before and/or after your sleep study.
- There's little work on your part: The device automatically sends your results to the licensed sleep doctor, so you don't have to worry about waiting for a telehealth appointment after finishing your study.
- It's lightweight, wearable, and easy to setup: Plus, it's pretty non-invasive, especially since you get to take the test from the comfort of your own bed.
What Are the Drawbacks of an At-Home Sleep Study?
- No technician to monitor the study: Leads or sensors may come off during sleep and fail to record important information. With at-home tests, there’s no technician available to place them again or check that the device is working properly.
- More limited data: At-home tests can’t truly track sleep or arousals because they don’t measure brain waves. This may lead to underestimating the impact that breathing events have on sleep during the study. For the same reason, these tests can’t be used to diagnose other sleep disorders.
- Less accurate AHI measurement: At-home devices use different methods of calculating the AHI than a lab sleep test does. As a result, the reported AHI number may be lower than it actually is. This can lead to a missed sleep apnea diagnosis or an underrepresentation of the degree of sleep apnea.
- Equipment differences: There is no set standard regarding which sensors or algorithms to use across devices, which can cause a lack of uniform results. The SCOPER categorization system has been proposed as a way to evaluate how well home sleep apnea tests measure sleep, cardiovascular activity, oximetry, position, effort, and respiration. However, not all HSATs adhere to this criteria.
- Doesn't completely rule out sleep apnea: A home sleep study can detect signs of obstructive sleep apnea, but it may miss milder cases or other variations, especially if symptoms don’t occur every night. That means a negative result doesn’t always guarantee you don’t have sleep apnea, and further testing might still be needed.
- You might still have other sleep issues: At-home tests are focused on identifying sleep apnea and don’t track other potential issues like insomnia, narcolepsy, or periodic limb movement disorder. If you’re still experiencing fatigue, poor sleep quality, or other symptoms, it’s worth discussing additional testing with your doctor.
WatchPAT ONE vs. Competitors
| WatchPAT ONE | Daybreak | In-Lab Testing | |
| Price | $189 | $160 | $1,000 – $10,000 |
| Measurements | Heart rate, blood oxygen level, pulse volume changes, snoring, body movement, chest movement, body positioning/movement | A variety of bodily metrics, including blood oxygen levels, snoring, and respiratory effort | In person sleep studies are usually more invasive and use more sensors to measure things like brain waves, heart activity, breathing, respiratory activity |
| Nights to Wear Device | 1 | 3 | 1 |
| Diagnosis Process | Meet with a doctor virtually, wear sensor for one night, and then meet with your doctor again to go over your results. From there, you may be able to talk through next steps and receive a prescription for PAP treatment if needed. | Wear the sensor for two nights, then a doctor will review your results and provide a diagnosis | Like an at-home study, a doctor will review your data and follow up with you to go over your results |
| Insurance Coverage | No, but FSA/HSA eligible | Yes | Yes |
Is an At-Home Sleep Study Right for You?
At-home sleep studies can be a good alternative for diagnosing sleep apnea for those who:
- May have moderate or severe obstructive sleep apnea, based on their symptoms
- Do not have certain medical conditions, like chronic obstructive pulmonary disease (COPD) or congestive heart failure
- Do not have insomnia or need to be evaluated for another sleep disorder, like sleepwalking or narcolepsy
Taking a sleep apnea test at-home might be a good option for you if you have a hectic schedule, want results right away, or simply don’t like the idea of sleeping in a sleep lab.
However, since different sleep disorders share common features, having a sleep study in a lab is the best way to distinguish sleep apnea from other sleep disorders.
What Is the Best At-Home Sleep Study?
Several devices on the market may be used for at-home sleep studies. For adults over the age of 18, the WatchPAT is an FDA-cleared home sleep test and one of the more advanced options on the market.
It works by measuring seven different physiological variables, including peripheral arterial tone (PAT), body position, heart rate, oxygen saturation, body movement, snoring, and chest motion. And because it relies on PAT measurements, it doesn’t require a chest band or nasal tubes. It consists simply of a device that fits on your wrist like a watch, a finger sensor, and a chest sensor.
Your WatchPAT will be delivered to you in the mail. After you complete the study, you’ll attend a video consultation with a sleep specialist to review your results, develop a treatment plan, and get a prescription if needed.
How Do You Prepare for an At-Home Sleep Study?
Preparing for an at-home sleep study is simple, but a little planning can help ensure accurate results:
- Follow your usual bedtime routine: Stick to your normal sleep and wake times to capture a typical night of sleep.
- Avoid caffeine and alcohol before bed: These substances can affect your sleep quality and interfere with test accuracy.
- Charge or insert batteries in your device: Make sure the testing device is fully powered and ready to use.
- Read the instructions carefully: Your home sleep test will come with a guide or video tutorial—review it thoroughly before starting.
- Attach the equipment properly: Most tests include sensors that go on your finger, chest, and under your nose. Proper placement is key to getting good data.
- Sleep in a comfortable environment: Choose a quiet, dark, and cool room to promote uninterrupted sleep.
By preparing ahead of time, you help your provider get the clearest picture of your nighttime breathing and overall sleep health.
Frequently Asked Questions
The WatchPAT ONE is a device that helps you take an at-home sleep study. It consists of three sensors that are worn on your finger, wrist, and chest to help you find out if you have sleep apnea, ultimately bringing you closer to treatment and healthy, restful sleep.
Once you order and set up your WatchPAT ONE, you can set up a telehealth appointment with a licensed sleep doctor to talk through any concerns or symptoms you're having. From there, you should prepare for your at-home sleep study by following your typical nighttime routine, making sure the device is charged and ready to go, and reading the instructions carefully. Put the sensors on your finger, wrist, and chest, which measure peripheral arterial tone, heart rate, blood oxygen, actigraphy, body position, snoring, and chest motion.
Once you've completed the study, you'll receive your tests results (your AHI score) in a follow-up email, and they'll automatically be sent to your licensed sleep doctor. You can meet once more with your doctor to talk through next steps. If you've been diagnosed with sleep apnea, you may be prescribed PAP therapy.
The WatchPAT ONE is fairly accurate as long as you put everything on as the instructions direct. The sensors provide multiple readings, like your heart rate, blood oxygen level, and sleeping positions throughout the night.
The WatchPAT ONE isn't covered by insurance, but it's HSA and FSA-approved.
The cost of an at-home sleep study typically ranges from $100 to $500—sometimes up to $1,000—depending on the provider and the equipment used. Some providers include follow-up consultations and analysis in the total cost.
It depends. Many insurance plans, including Medicare, cover at-home sleep studies only if deemed medically necessary and prescribed by a doctor. Coverage varies, so it’s best to check with your insurance provider to confirm what’s included and whether prior authorization is needed.
You should aim for at least 4 to 6 hours of sleep during the study. This duration helps ensure the device collects enough data to provide an accurate assessment of your breathing and sleep patterns.
Yes, you can get up to use the bathroom. Just be careful not to dislodge any sensors or equipment, and make sure everything is securely in place when you return to bed. Most devices will continue recording as long as they stay properly connected.